Project coordination and dissemination
Led by the Groupe de Recherche Action en Santé (GRAS), this work Package is in charge of the overall coordination and management of the project. The specific objectives are then, to:
- provide overall management to the project ;
ensure effective coordination of the scientific program and communication within and beyond the consortium ;
ensure timely disbursement of budget ;
ensure implementation of the project activities in respect of the work plan;
established good communication channels between partners and stakeholders ;
inform the scientific community, regulatory authorities and the broad population on the progress in malaria vaccine development ;
To ensure successful implementation and conduct of the program, the coordinating site, GRAS, is being assisted by key governing and advisory bodies, namely the Project Steering Committee (PSC) , the General Assembly (GA), and the External Independent Scientific Advisory Group (EISAG).


CHMI trials in Europe & Africa
This WP co led by Ifakara Health Institute Trust (IHI), Tanzania and Eberhard Karls Universität Tübingen (EKUT), Germany, aims to conduct controlled human malaria infection (CHMI) trials in Africa and Europe during the first two years of the program to rapidly test and compare targeted malaria vaccine candidates and down-select the most promising one for phase 2b trial in endemic areas. Other collaborators involved are KEMRI-WT/Kenya (UOXF) for immunological evaluations. The specific objectives of this WP are to:
compare efficacy of most promising pre-erythrocytic malaria vaccine candidates in malaria-naïve adults against standardized CHMI with PfSPZ Challenge strain 7G8 ;
compare safety and tolerability of tested vaccine candidates ;
compare immunogenity of tested vaccine candidates ;
identify and select lead malaria vaccine candidates for trialing in the multicentre phase 2b trial in African children ;
evaluate safety and immunogenicity of blood-stage malaria vaccine candidates in adults and children residing in malaria endemic countries ;
evaluate the efficacy of blood-stage malaria vaccines candidate against heterologous CHMI administered by DVI to adults residing in endemic countries.
Phase 2b trials in Africa (VALiT)
The VALiT trial is led by Centre de Recherches Médicales de Lambaréné (CERMEL) and co-led by Fundação Manhiça, Mozambique (CISM/FM). It is a multicentre, double-blind, randomized, controlled phase 2 trial of lead malaria vaccine candidates that will be conducted in African children under exposure to natural infection with P. falciparum. More than 4 candidates (max. 2 pre-eryhrthrocytic and max. 2 blood stage immunogens) may be selected and tested in the trial sites of Gabon, Burkina Faso and Mozambique. The study specifically aims to:
assess safety and tolerability of vaccine candidates in children;
determine efficacy of vaccine candidates in children under natural exposure to P. falciparum infections;
characterize immune response;
identify lead malaria vaccine candidate (s) for advanced clinical development.

Quality Assurance & Networking
Quality Assurance and networking is dedicated to ensure high-quality trial outcomes by implementing quality assurance systems in all trial sites and closely monitoring study activities and safety data and the sustained development of CHMI capacities and CHMI platform that ensures vaccine development activities beyond the project. Trial specific DSMBs are established to provide oversight for CHMI trials and field efficacy trials.
The sponsors with delegation to EVI and the CROs (selected by EVI) ensures that the studies and trials are performed and data are generated, documented and reported in compliance with ICH GCP and applicable regulatory requirements, and are standardised across the sites.
This work package is led by the European Vaccine Initiative EWIV (EVI) in collaboration with the clinical trial sites. Its specific objectives are, to:
ensure the implementation of quality assurance system in all clinical trial sites;
develop clinical monitoring plan for CHMI model studies;
develop clinical monitoring plan for field trials;
ensure implementation of a qualified and experienced independent Data Safety Monitoring Board in all clinical trials ;
ensure harmonisation of clinical study procedures and laboratory assays across sites;
ensure networking within and outside the consortium.
Data management
This work package, led by Luxembourg Institute of Health (LIH) and co led by GRAS, aims to ensure that data generated in the programme are handled appropriately in compliance with current regulations. The specific objectives are to:
develop a Data Management Plan (DMP) that will describe the overall vision/approach for managing the data generated during/after the lifespan of the programme;
set up a Data Management System that will be used for the data management at the second stage of the programme;
ensure data management comply with current regulations.
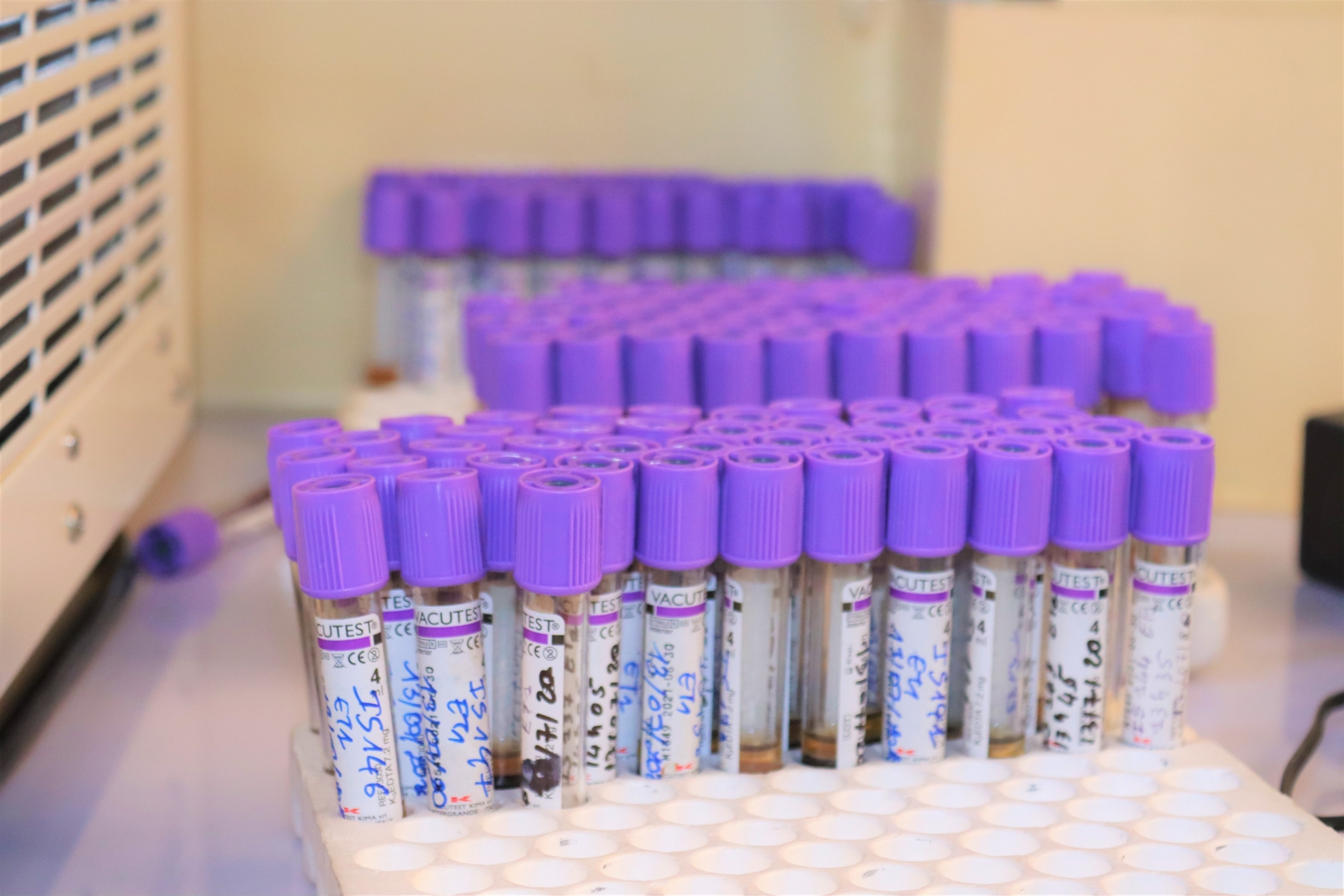
Immunology
WP6 Immunology is dedicated to assess immunogenicity of the vaccine candidates in the different populations of the trials, and potentially, identify immune correlates of protection.
The Chancellor, the Masters and the Scholars of the University of Oxford (UOXF; KEMRI–Welcome Trust Research Programme) and the (LUMC) are co leading this WP with contributions of the trial sites. The specific objectives are to:
characterise the immune responses induced by the vaccine candidates tested in WP2 and WP3;
define immune correlates of protection in CHMI trials;
characterize and compare immune response to the same vaccine antigen amongst malaria-naives and African children;
characterize the immune responses to malaria vaccine candidates within the consortium portfolio.
Capacity building and baseline study
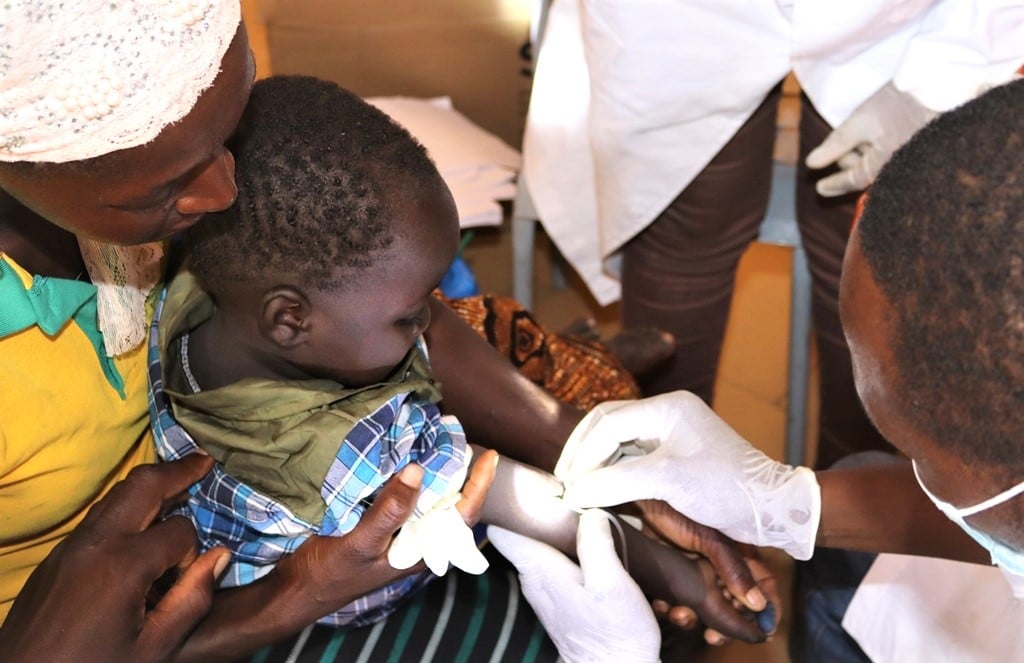
Groupe de Recherche Action en Santé (GRAS) is leading the WP7 which comprises Capacity building and baseline studies. The baseline study (BLOOMy) is in fact a malaria morbidity assessment in the three foreseen phase 2b trial sites. The specific objectives of the WP are to:
assess capabilities and gaps within the partner institutions and identify those that can be shared with others and that needs strengthening;
generate baseline malaria incidence data to informe the field trials design;
train and support MSc, PhD student, medical students and post-doctoral researchers.



